The world of genetic engineering has taken another leap forward with the introduction of CRISPR experiment kits designed specifically for editing fluorescent protein genes in yeast. These kits, now available to researchers and educators alike, provide a hands-on approach to understanding the revolutionary CRISPR-Cas9 technology. By targeting the genes responsible for fluorescence in yeast, users can witness firsthand the power and precision of genome editing.
CRISPR technology has transformed the field of molecular biology, offering unprecedented control over genetic material. The ability to make precise cuts in DNA at specific locations has opened up new possibilities for research, medicine, and biotechnology. The new experiment kits focus on yeast as a model organism, leveraging its well-characterized genome and rapid growth to demonstrate CRISPR's capabilities in a classroom or laboratory setting.
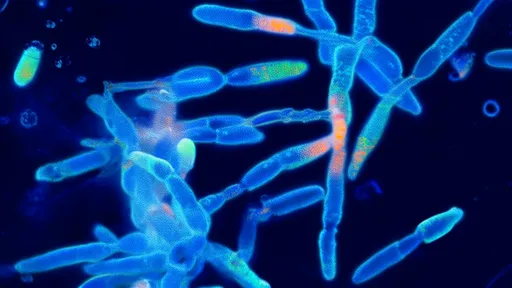
One of the most exciting aspects of these kits is their focus on fluorescent proteins. Yeast strains engineered to produce fluorescent proteins are not only visually striking but also serve as excellent tools for teaching genetic concepts. When students or researchers successfully edit these genes, the changes are immediately visible under UV light, providing instant feedback on their experiments.
The safety considerations surrounding these kits have been carefully addressed. While CRISPR technology is powerful, the specific applications in these educational kits are designed to be completely safe. The modifications are limited to non-pathogenic yeast strains, and the targeted genes are those involved in harmless traits like fluorescence. This makes the kits suitable for use in high school and undergraduate laboratory courses where safety is paramount.
Beyond educational applications, these CRISPR kits represent an important step in democratizing genetic engineering technology. By making sophisticated tools accessible and affordable, they lower the barrier to entry for students and citizen scientists interested in biotechnology. This accessibility could help cultivate the next generation of genetic engineers and molecular biologists.
The experimental procedure typically begins with preparing the yeast cells and the CRISPR-Cas9 components. Users then design guide RNAs that target specific sequences in the fluorescent protein genes. After introducing these components into the yeast cells, the CRISPR system goes to work, making precise edits to the DNA. Within days, colonies begin to show altered fluorescence patterns, demonstrating successful genome editing.
What makes these kits particularly valuable is their ability to bridge the gap between theoretical knowledge and practical application. Many students learn about CRISPR in textbooks or lectures, but actually performing the procedure provides a much deeper understanding of how the technology works. The visual nature of the fluorescent protein system makes the results immediately apparent, reinforcing the learning experience.
As CRISPR technology continues to advance, its applications in education are becoming increasingly sophisticated. These yeast fluorescence editing kits represent just one example of how cutting-edge science can be adapted for instructional purposes. Future developments may include more complex editing scenarios or the incorporation of additional fluorescent markers to demonstrate different genetic concepts.
The implications of this technology extend beyond the classroom. By familiarizing students with CRISPR early in their academic careers, these kits help prepare the next generation of scientists for the challenges and opportunities of modern genetic engineering. As the field continues to evolve, hands-on experience with tools like these will become increasingly valuable for aspiring biologists.
Looking ahead, we can expect to see continued innovation in educational CRISPR kits. While the current focus is on visible markers like fluorescent proteins, future versions might incorporate more complex traits or even multi-gene editing scenarios. The success of these yeast-based kits may also inspire similar products using other model organisms, further expanding the possibilities for hands-on genetics education.
For educators considering incorporating these kits into their curriculum, the benefits are clear. They provide a safe, engaging, and effective way to teach modern genetic engineering techniques. The visual nature of the experiments helps students grasp abstract concepts, while the hands-on nature of the work develops important laboratory skills. As genetic engineering becomes increasingly important across multiple scientific disciplines, early exposure to these technologies becomes ever more valuable.
The development of these CRISPR experiment kits reflects a broader trend in science education: the move toward more interactive, experiential learning. Rather than simply reading about scientific breakthroughs, students can now participate in them, albeit on a smaller scale. This shift has the potential to inspire more students to pursue careers in biotechnology and related fields.
As with any new educational technology, there will be challenges to overcome. Some institutions may need to upgrade their laboratory facilities to accommodate these experiments, and instructors will require training to effectively teach the material. However, the long-term benefits of giving students hands-on experience with cutting-edge genetic tools far outweigh these initial hurdles.
The CRISPR yeast fluorescence editing kits represent an exciting development at the intersection of science and education. By making advanced genetic engineering techniques accessible to students, they help demystify a technology that will undoubtedly shape the future of biology and medicine. As these kits become more widely adopted, they may fundamentally change how we teach genetics in the 21st century.

By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025
By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025

By /Aug 7, 2025